Table of Contents
SN1 Reaction
The SN1 reaction is a nucleophilic substitution reaction where the rate-determining step is unimolecular. It is a type of organic substitution reaction. SN1 stands for substitution nucleophilic unimolecular. Thus, the rate equation (which states that the SN1 reaction is dependent on the electrophile but not on the nucleophile) holds in situations where the amount of the nucleophile is far greater than the amount of the carbocation intermediate.
This reaction involves the formation of a carbocation intermediate. It is generally seen in the reactions of tertiary or secondary alkyl halides with secondary or tertiary alcohols under strongly acidic or strongly basic conditions. The SN1 reaction is often referred to as the dissociative mechanism in inorganic chemistry.
SN1 Reaction Mechanism
SN1 reaction mechanism follows a step-by-step process wherein first, the carbocation is formed from the removal of the leaving group. Then the carbocation is attacked by the nucleophile. Finally, the deprotonation of the protonated nucleophile takes place to give the required product. The rate-determining step of this reaction depends purely on the electrophilicity of the leaving group and is not impacted at all by the nucleophile
SN1 indicates a substitution, nucleophilic, unimolecular reaction, described by the expression
rate = k [R–LG].
This implies that the rate-determining step of the mechanism depends on the decomposition of a single molecular species.
This pathway is a multi-step process with the following characteristics:
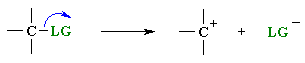 |
Step 1: slow loss of the leaving group (LG), to generate a carbocation intermediate, then | |
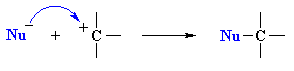 |
Step 2: rapid attack of a nucleophile on the electrophilic carbocation to form a new bond |
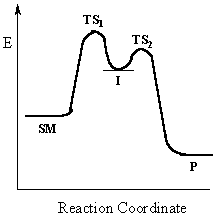 |
Multi-step reactions have intermediates and several transition states (TS).In an SN1 there is the loss of the leaving group generates an intermediate carbocation which is then undergoing a rapid reaction with the nucleophile. |
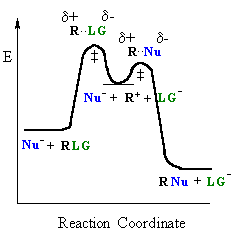 |
| General case | SN1 reaction |
Reactivity order :
(CH3)3C- > (CH3)2CH- > CH3CH2– > CH3–
3º > 2º >> 1º >> > zero order
In an SN1 reaction, the rate determining step is the loss of the leaving group to form the intermediate carbocation. The more stable the carbocation is, the easier it is to form, and the faster the SN1 reaction will be. Some students fall into the trap of thinking that the system with the less stable carbocation will react fastest, but they are forgetting that it is the generation of the carbocation that is the rate determining.
Effect of Solvent
- A solvent that can facilitate the formation of the carbocation intermediate will speed up the rate determining step of the SN1 reaction.
- The preferred solvents for this type of reaction are both polar and protic.
- The polar nature of the solvent helps to stabilize ionic intermediates whereas the protic nature of the solvent helps solvate the leaving group.
Examples of solvents used in SN1 reactions include water and alcohols. These solvents also act as nucleophiles.
Leaving Group
The only event in the rate-determining step of the SN1 is breaking the C–LG bond. Therefore, there is a very strong dependence on the nature of the leaving group, the better the leaving, the faster the SN1 reaction will be.
Nucleophile
Since the nucleophile is not involved in the rate-determining step, the nature of the nucleophile is unimportant in an SN1 reaction. However, the more reactive the nucleophile, the more likely an SN2 reaction becomes.
Solvent
Polar solvents which can stabilize carbocations which can favour the SN1 reaction (e.g. H2O, ROH)
Stereochemistry of SN1 Reaction
The carbocation intermediate formed in step 1 of the SN1 reaction mechanism is an sp2 hybridized carbon. Its molecular geometry is trigonal planar, therefore allowing for two different points of nucleophilic attack, left and right.
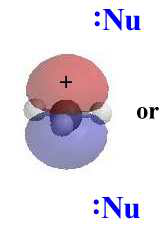 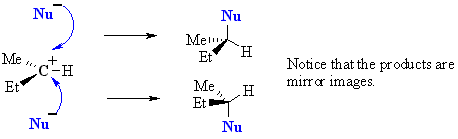 In an SN1, the nucleophile attacks the planar carbocation. Since there is an equally probability of attack on each face there will be a loss of stereochemistry at the reactive center as both products will be observed. |
If the reaction takes place at a stereocenter and if neither avenue for nucleophilic attack is preferred, the carbocation is then attacked equally from both sides, yielding an equal ratio of left and right-handed enantiomers.







