Table of Contents
Introduction Of Emulsions
Emulsion is a heterogeneous system consisting of one immiscible liquid dispersed in another in the form of droplets whose diameter, in general, exceeds 0.1 microns. Such a system possesses minimal stability because the droplets quickly coalesce and the two liquids get separated. The stability of the emulsion is increased by adding another substance known as the emulsifying agent or emulsifiers. The liquid droplets are generally known as the dispersed phase or internal phase, whereas the liquid in which they are dispersed is known as the continuous phase, dispersion medium, or external phase. Emulsion increases the stability of many drugs that are unstable in an aqueous solution. It improves the taste of objectionable medicinal agents and makes them more acceptable. It also improves penetration and spreadability. Emulsions enhance the rate and extent of absorption through the alimentary canal, improve the solubility of poorly water-soluble drugs, and can prolong drug action. Examples are liquid paraff in emulsion, cod liver oil emulsion.
There are two types of emulsion:
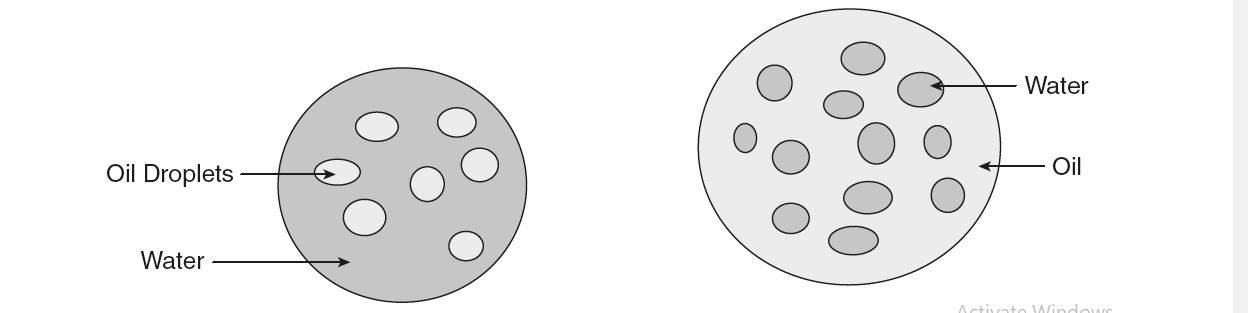
1. Oil in water (O/W) emulsions: The oil (internal or dispersed) phase is dispersed as droplets throughout the aqueous phase (external or continuous phase or dispersion medium). Oil is the dispersed phase and water is the dispersion medium.
2. Water in oil (W/O) emulsions: The internal phase is composed of water droplets and the external phase is nonaqueous. Water is the dispersed phase and oil is the dispersion medium. In general, all oral emulsions tend to be oil-in-water as the oily phase is usually less pleasant to take and more difficult to flavor. The differences between O/W and W/O emulsions are provided
The major use of emulsions is as cream formulations (for external application). However, they may
also be administered intravenously, rectally, or orally. Emulsions are physically unstable and the Vario.
| Oil in Water Emulsion (O/W) | Water in Oil Emulsion (W/O) |
| Oil is the dispersed phase and water is the dispersion medium | Water is the dispersed phase and oil is the dispersion medium. |
| They are nongreasy and easily washed from the skin surface. | They are greasy and not easily washed by water. |
| They are used externally to provide a cooling effect, for example, vanishing cream. | They are used externally to prevent evaporation of moisture from the surface of the skin, for example, cold cream |
| Water-soluble drugs are more quickly released from O/W emulsions. | Oil-soluble drugs are more quickly released from W/O emulsions |
| They are preferred for oral formulations as the taste of oils can be masked. | They are preferred for topical preparations such as creams |
| O/W emulsions give a positive conductivity test since the external phase is water, which is a good conductor of electricity. | W/O emulsions do not give a positive conductivity test since the external phase is oil, which is a poor conductor of electricity. |
Micro-emulsions
Clear dispersions of oil in water or water in oil are called microemulsions. The disperse phase globules are very small, ranging in diameter from 100A° to 600A°. These emulsions appear as transparent solutions and are more acceptable physically as compared to conventional emulsions.
Self-emulsifying systems
These are anhydrous products that when added to excess water form emulsions spontaneously without requiring too much agitation. These systems are known as self-emulsifying drug delivery systems (SEDDS) and can be formulated by the use of a combination of hydrophilic and lipophilic surfactants.
Complex emulsions (multiple emulsions)
1. W/O/W emulsions: An oil droplet enclosing a water globule may be dispersed in water to give W/O/W emulsions; see Fig. 6.2(a).
2. O/W/O emulsions: A water droplet enclosing an oil globule may be dispersed in oil to give O/W/O emulsions; see Fig. 6.2(b).
Advantages and Disadvantages of emulsions as Dosage forms
The following are the advantages of using emulsions as dosage forms:
1. Unpalatable oils can be administered in a palatable form.
2. The aqueous phase is easily flavored.
3. The taste of oils can be masked.
4. Absorption is faster when compared to solid dosage forms.
5. It is possible to include two incompatible ingredients, one in each phase of the emulsion.
6. Emulsions of the O/W type are administered orally for the following purposes:
(a) Disguise the taste or oiliness of medicinal oils such as liquid paraffin and cod liver oil. The aqueous continuous phase, which will be pleasantly flavored, isolates the unpleasant disperse phase from the tongue and, if the patient wishes, allows the residue of the dose to be rinsed easily from the mouth with water.
(b) To improve the absorption of oils
(c) Sometimes O/W emulsions of nutritive oils and fats are administered intravenously to patients who are unable to ingest food in the normal way.
7. Semisolid emulsions are O/W (vanishing creams) or W/O (cold creams). O/W emulsions are preferred over W/O emulsions. They can be easily rubbed into the skin and can be easily removed by washing
The following are the disadvantages of using emulsions as dosage forms:
1. Calculation of primary emulsion formulae and technical expertise are needed for the manufacture of stable emulsions.
2. A measuring device is needed for administration.
3. Emulsions require shaking before use to ensure uniformity of dose.
4. If the preparation is not shaken well, the accuracy of the dose is likely to be less when compared to solutions.
5. Improper storage conditions can affect the disperse system.
6. Since emulsions are stored in glass or plastic containers, they are bulky, difficult to transport, and prone to container breakages.
7. Microbial contamination of emulsions can lead to cracking.