Table of Contents
ELISA
ELISA is an antigen-antibody reaction. In 1971, ELISA was introduced by Peter Perlmann and Eva Engvall at Stockholm University in Sweden. It is a common laboratory technique which is usually used to measure the concentration of antibodies or antigens in the blood.
ELISA is a plate-based assay technique which is used for detecting and quantifying substances such as peptides, proteins, antibodies, and hormones. An enzyme conjugated with an antibody reacts with colorless substrate to generate a colored product. Such substrate is called chromogenic substrate. A number of enzymes have been used for ELISA such as alkaline phosphatase, horse radish peroxidase and beta-galactosidase. Specific substrate such as ortho-phenyl diamine dihydrochloride (for peroxidase), para nitrophenyl phosphate (for alkaline phosphatase) are used which are hydrolyzed by the above enzymes to give colored end product.
Principle
ELISAs are typically performed in 96-well polystyrene plates. The serum is incubated in a well, and each well contains a different serum. A positive control serum and a negative control serum would be included among the 96 samples being tested. Antibodies or antigens present in serum are captured by corresponding antigen or antibody-coated onto the solid surface. After some time, the plate is washed to remove serum and unbound antibodies or antigens with a series of wash buffer. To detect the bound antibodies or antigens, secondary antibody that are attached to an enzyme such as peroxidase or alkaline phosphatase are added to each well. After an incubation period, the unbound secondary antibodies are washed off. When a suitable substrate is added, the enzyme reacts with it to produce a color. This color produced is measurable as a function or quantity of antigens or antibodies present in the given sample. The intensity of color/ optical density is measured at 450nm. The intensity of the color gives an indication of the amount of antigen or antibody.
Types of ELISA

Frequently there are 4 types of ELISA on the basis of binding structure between the Antibody and Antigen.
- Direct ELISA
- Indirect ELISA
- Sandwich ELISA
- Competitive ELISA
Direct ELISA

In direct ELISA, only an enzyme-labeled primary antibody is used, meaning that secondary antibodies are not needed. The enzyme-labeled primary antibody “directly” binds to the target (antigen) that is immobilized to the plate (solid surface). Next, the enzyme linked to the primary antibody reacts with its substrate to produce a visible signal that can be measured. In this way, the antigen of interest is detected.
- Fast and simple protocol
- Less specific since you are only using 1 antibody.
- Potential for high background if all proteins from a sample are immobilized in well.
Indirect ELISA
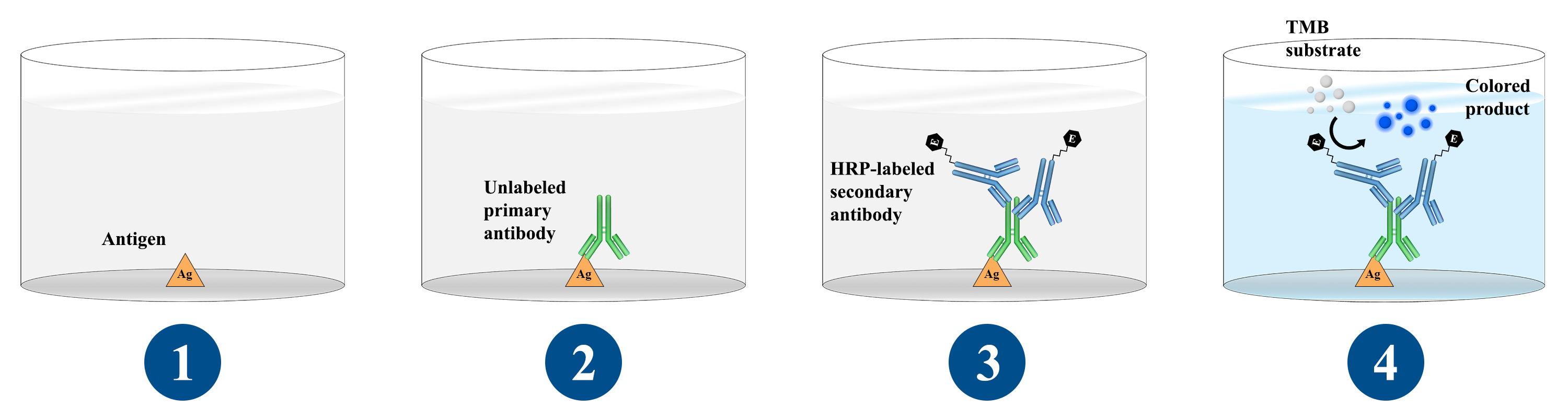
Antibody can be detected or quantitatively determined by indirect ELISA. In this technique, antigen is coated on the microtiter well. Serum or some other sample containing primary antibody is added to the microtiter well and allowed to react with the coated antigen. Any free primary antibody is washed away and the bound antibody to the antigen is detected by adding an enzyme conjugated secondary antibody that binds to the primary antibody. Unbound secondary antibody is then washed away and a specific substrate for the enzyme is added. Enzyme hydrolyzes the substrate to form colored products. The amount of colored end product is measured by spectrophotometric plate readers that can measure the absorbance of all the wells of 96-well plate.
Procedure of Indirect ELISA
- Coat the micro titer plate wells with antigen.
- Block all unbound sites to prevent false positive results.
- Add sample containing antibody (e.g. rabbit monoclonal antibody) to the wells and incubate the plate at 37°c.
- Wash the plate, so that unbound antibody is removed.
- Add secondary antibody conjugated to an enzyme (e.g. anti- mouse IgG).
- Wash the plate, so that unbound enzyme-linked antibodies are removed.
- Add substrate which is converted by the enzyme to produce a colored product.
- Reaction of a substrate with the enzyme to produce a colored product.
Advantages
- Increased sensitivity, since more than one labeled antibody is bound per primary antibody.
- A wide variety of labeled secondary antibodies are available commercially.
- Maximum immunoreactivity of the primary antibody is retained because it is not labeled.
- Versatile because many primary antibodies can be made in one species and the same labeled secondary antibody can be used for detection.
- Flexibility, since different primary detection antibodies can be used with a single labeled secondary antibody.
- Cost savings, since fewer labeled antibodies are required.
- Different visualization markers can be used with the same primary antibody.
Disadvantages
- Cross-reactivity might occur with the secondary antibody, resulting in nonspecific signal.
- An extra incubation step is required in the procedure.
Sandwich ELISA
Antigen can be detected by sandwich ELISA. In this technique, antibody is coated on the microtiter well. A sample containing antigen is added to the well and allowed to react with the antibody attached to the well, forming antigen-antibody complex. After the well is washed, a second enzyme-linked antibody specific for a different epitope on the antigen is added and allowed to react with the bound antigen. Then after unbound secondary antibody is removed by washing. Finally substrate is added to the plate which is hydrolyzed by enzyme to form colored products.
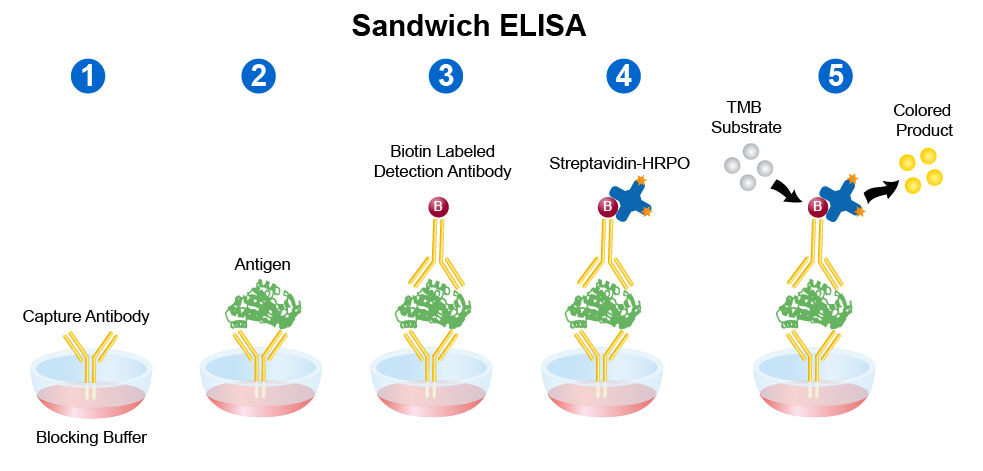
Procedure of sandwich ELISA
- Prepare a surface to which a known quantity of antibody is bound.
- Add the antigen-containing sample to the plate and incubate the plate at 37°c.
- Wash the plate, so that unbound antigen is removed.
- Add the enzyme-linked antibodies which are also specific to the antigen and then incubate at 37°c.
- Wash the plate, so that unbound enzyme-linked antibodies are removed.
- Add substrate which is converted by the enzyme to produce a colored product.
- Reaction of a substrate with the enzyme to produce a colored product.
Advantages
- High specificity, since two antibodies are used the antigen is specifically captured and detected.
- Suitable for complex samples, since the antigen does not require purification prior to measurement.
- Flexibility and sensitivity, since both direct and indirect detection methods can be used.
Competitive ELISA
This test is used to measure the concentration of an antigen in a sample.
In this test, antibody is first incubated in solution with a sample containing antigen. The antigen-antibody mixture is then added to the microtitre well which is coated with antigen. The more the antigen present in the sample, the less free antibody will be available to bind to the antigen-coated well. After the well is washed, enzyme conjugated secondary antibody specific for isotype of the primary antibody is added to determine the amount of primary antibody bound to the well. The higher the concentration of antigen in the sample, the lower the absorbance.

Procedure
- Antibody is incubated with a sample containing the antigen.
- Antigen-antibody complex are added to the microtitre well which are pre-coated with the antigen.
- Wash the plate to remove unbound antibody.
- Enzyme linked secondary antibody which is specific to the primary antibody is added.
- Wash the plate, so that unbound enzyme-linked antibodies are removed.
- Add substrate which is converted by the enzyme into a fluorescent signal.
Advantages
- High specificity, since two antibodies are used.
- High sensitivity, since both direct and indirect detection methods, can be used.
- Suitable for complex samples, since the antigen does not require purification prior to measurement.
Advantages and disadvantages of each ELISA type
| Advantages | Disadvantages | |
| Direct ELISA | 1. Simple protocol, time-saving, and reagents-saving.
2. No cross-reactivity from secondary antibody. |
1. High background.
2. No signal amplification, since only a primary antibody is used and a secondary antibody is not needed. 3. Low flexibility, since the primary antibody must be labeled. |
| Indirect ELISA | 1. Signal amplification, since one or more secondary antibodies can be used to bind to the primary antibody.
2. High flexibility, since the same secondary antibody can be used for various primary antibodies. |
1. Complex protocol compared with direct ELISA.
2. Cross-reactivity from secondary antibody. |
| Sandwich ELISA | 1. High flexibility.
2. High sensitivity. 3. High specificity, since different antibodies bind to the same antigen for detection. |
1. The antigen of interest must be large enough so that two different antibodies can bind to it at different epitopes.
2. It’s sometimes difficult to find two different antibodies that recognize different epitopes on the antigen of interest and cooperate well in a sandwich format. |
| Competitive ELISA | 1. High flexibility.
2. High sensitivity. 3. Best for the detection of small antigens, even when they are present in low concentrations. |
1. Relatively complex protocol.
2. Needs the use of inhibitor antigen. |
Application of ELISA
- Presence of antigen or the presence of antibody in a sample can be evaluated.
- Determination of serum antibody concentrations in a virus test.
- Used in food industry when detecting potential food allergens.
- Applied in disease outbreaks- tracking the spread of disease e.g. HIV, bird flu, common, colds, cholera, STD etc.
References
https://www.laboratoryinsider.com/2019/04/elisa-principle-types-and-applications.html.
https://microbiologynotes.com/author/piku/page/6/.
https://microbiologynotes.com/elisa-principle-types-and-applications/.
https://www.labtestsguide. com/elisa
https://www.bio.davidson.edu/courses/genomics/method/ELISA.html.
https://enzymeimmunoassay.wordpress.com/prinicple/.
https://enzymeimmunoassay.wordpress.com/types/.
https://vulms.vu.edu.pk/Courses/BT102/Downloads/ELISA%20technique.docx.
https://quizlet.com/tr/456070442/microbiology-immunological-techniques-flash-cards/.
https://www.cusabio.com/c-20659.html.
https://probreaking.org/docs/direct-elisa-principle-108857.
https://quizlet.com/gb/238518076/elisa-flash-cards/.
https:// www.scribd.com/document/405565769/Elisa







